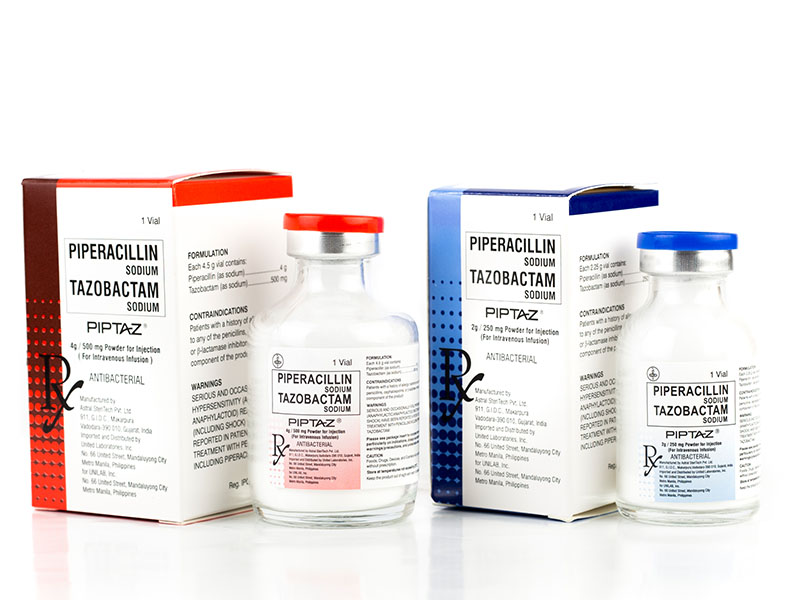
| Each 2.25 g vial contains: | |
| Piperacillin (as sodium) | 2 g |
| Tazobactam (as sodium) | 250 mg |
| Each 4.5 g vial contains | |
| Piperacillin (as sodium) | 4 g |
| Tazobactam (as sodium) | 500 mg |
Piperacillin Sodium + Tazobactam Sodium
Piptaz®
Prescription Drug
Indications
For the treatment of the following moderate to severe infections caused by piperacillin-resistant, piperacillin/tazobactam-susceptible, and β-lactamase producing strains of susceptible microorganisms:
Adults and Adolescents
- Lower respiratory tract infections including severe community-acquired pneumonia and healthcare-associated pneumonia
(Note:Healthcare-associated pneumonia caused by Pseudomonas aeruginosa should be treated in combination with an aminoglycoside)
- Uncomplicated and complicated skin and skin structure infections including cellulitis, cutaneous abscesses, and acute ischemic/diabetic foot infections
- Intraabdominal infections including appendicitis (complicated by rupture and abscess) and peritonitis
- Complicated and uncomplicated urinary tract infections
- Gynecologic infections including postpartum endometritis or pelvic inflammatory disease
- Bone and joint infections
- Bacterial sepsis
- Bacterial infections in neutropenic patients
(Note: Full therapeutic doses of piperacillin/tazobactam plus an aminoglycoside should be used)
- Polymicrobic infections including those where aerobic and anaerobic organisms are suspected (intra-abdominal, skin and skin structure, upper and lower respiratory tract, gynecological).
Children
- Complicated intra-abdominal infections in children ≥ 2 months old
- Bacterial infections in febrile neutropenic children 2 to 12 years old
Dosage and Administration
For more information on safety, precaution and other information about this product, please see the Product Insert.
Please consult your physician and other healthcare providers before taking any prescription medicines found on this site. The contents found on this page are for information purposes only. It should not be construed as a substitute for professional medical advise and should not be relied upon in that regard. You undertake to carefully read all product packaging and labels prior to use.
Caution: Food, Drugs, Devices and Cosmetics Act prohibits dispensing without prescription.


